|
Plants are autotrophs - they make their own food. They do this through the process of photosynthesis (light, making) - using the energy from light to make glucose. Plants combine carbon dioxide and water and enzymes (provided with energy from sunlight) turn this into glucose and oxygen.
|
All living things (including plants) need to have a source of energy in order to live. Glucose is the substrate for respiration, and with the help of enzymes it can be broken down, in the presence of oxygen, to release lots of ATP (energy), carbon dioxide and water (=aerobic reaction) or solely use glucose to release a small amount of ATP and other products (=anaerobic respiration).
|
Cell division is part of the cell cycle and is made up of two phases; interphase and the mitosis phase. The purpose of cell division is so that new cells can be made so that organisms can grow bigger or repair old/damaged cells. Cell division requires energy (therefore respiration). Cell division is essential for life!
|
To fully 'get' the above processes, you need to understand the following concepts
Movement of materials & enzymes
|
|
|
Materials move in and out of cells via
the Plasma Membrane
The structure of the plasma membrane is described as a phospholipid bilayer, made up of fatty acids containing hydrophilic (water-loving) heads and hydrophobic (water-hating) tails. Embedded in the membrane are proteins (described as a mosaic) and the membrane moves fluidly.
Plasma membranes are selectively permeable - there are small pores where substances can enter and exit the cell. Substances, such as water, can enter the cell without requiring the cell to use any energy whereas other substances, such as glucose or sodium, require the cell to use energy to facilitate the movement of these molecules through the membrane.
Plasma membranes are selectively permeable - there are small pores where substances can enter and exit the cell. Substances, such as water, can enter the cell without requiring the cell to use any energy whereas other substances, such as glucose or sodium, require the cell to use energy to facilitate the movement of these molecules through the membrane.
Movement of materials can be split up into two 'types' of movement
Passive transport and active transport
|
Passive transport is the movement of materials from a place of high concentration to a low concentration, down a concentration gradient.
"Going with the flow" Diffusion, facilitated diffusion and osmosis are all examples of passive transport. |
Active transport is the movement of materials from a place of low concentration to a high concentration, against a concentration gradient.
"Going against the flow" Carrier proteins and cytosis are both examples of active transport. |
Passive transport
-
Diffusion
-
Osmosis
-
Facilitated diffusion
-
Examples
<
>
The movement of particles - from an area of high concentration to an area of low concentration - until the particles are evenly distributed. The particles can move through gasses or liquids.
Factors affecting diffusion
|
The greater the concentration gradient (the difference in concentration between two areas/regions), the faster diffusion will occur
|
The greater the distance, the slower diffusion occurs. The shorter the distance, the faster diffusion occurs. This refers to the thickness of the membrane.
|
Thicker barriers slow the rate/speed of diffusion. Having pores in a cell membrane or barrier will enhance the rate of diffusion
|
The greater the temperature, the more energy particles have to move around. This means they will move down a concentration gradient faster if the temperature is higher
|
|
Osmosis is a special type of diffusion - it is directly related to water transport (rather than of any other particles). Water moves from an area of high concentration to an area of low concentration without any energy to assist the process. When the concentration of water molecules on both sides of the membrane is the same, the rate of water movement is the same either side. This is called equilibrium (equal).
|
Tonicity
- a way to measure the water potential gradient of two solutions (i.e. on either side of a cell membrane)
Check out this website - it is really helpful!
- a way to measure the water potential gradient of two solutions (i.e. on either side of a cell membrane)
Check out this website - it is really helpful!
Water potential
|
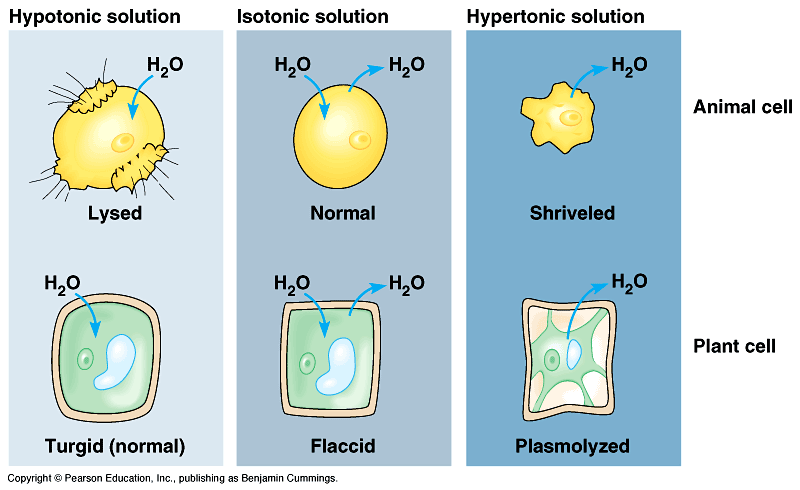
A measure of the tendency of water molecules to move from one place to another. Also called osmolarity. The water potential of a solution is always a comparison of that solution with another solution. One will have more water potential than another. The solution you have will either be hypertonic, hypotonic or isotonic compared to the other solution (often you will be comparing the inside of a cell to the outside of the cell). |
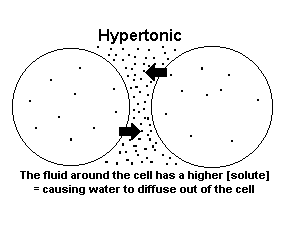
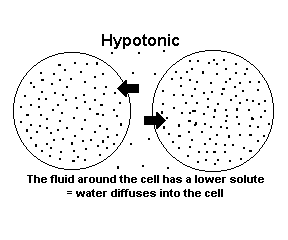
The dots below represent solute so keep that in mind as you read the diagrams!
|
When a solution is hypertonic, it contains a high amount of solute molecules (dissolved stuff) and a low amount of water molecules. In the picture above, the dots represent the solute - there is a high amount of solute in the solution outside of the cell so that solution is hypertonic to the cell. Inside the cell there is a higher concentration of water molecules compared to solute molecules. |
When a solution is hypotonic, it contains a high amount of water molecules and a low amount of solute molecules (dissolved stuff). In the picture above, the solution surrounding the cells is hypotonic in comparison to inside the cells. Inside the cell there is a higher concentration of solute molecules and a lower amount of water molecules. You could say the cell is hypertonic to the surrounding solution. |
When a solution is isotonic, the concentration of water molecules (or solute molecules) are the same in both environments, so there is no solution that is hyper- or hypo- tonic compared to the other (i.e. water doesn't move into or out of the cell to even out the concentrations). Water still moves between them, but it is at a constant rate and the cell does not swell or shrink. |
Implications
Words you need to remember
|
Animal cells - cell membrane, no cell wall
|
Plant cells - cell membrane and cell wall
|
Some molecules are too big to move through a cell membrane, even though they're travelling from a high to low concentration. Transport proteins are embedded in the cell membrane and in these proteins are channels to help with the diffusion of these larger molecules. As the molecules are moving from a high to low concentration, no energy is needed. Transport proteins are specific - they can only help one type of molecule through.
Changes in the concentration gradient don't affect the rate of diffusion but temperature does; if the temperature gets too hot the proteins (making up the protein channel) denature and diffusion won't be able to occur.
Active transport
-
Carrier proteins
-
Cytosis
-
Examples
<
>
To prevent excess water loss/gain, sometime solutes need to be pumped into/out of cells in order to maintain the right water balance. Other times, materials just need to be moved against the gradient in order to supply the cell with more of what it needs.
These molecules are moved across the cell membrane by active transport mechanisms, requiring energy as the movement of these molecules is against the natural concentration gradient. The 'pumping' of these molecules across the membrane is done by carrier proteins that are embedded in the membrane. ATP molecules (energy molecules) directly contribute to changing the shape of these carrier proteins, allowing them to open and close to let molecules in and out.
These molecules are moved across the cell membrane by active transport mechanisms, requiring energy as the movement of these molecules is against the natural concentration gradient. The 'pumping' of these molecules across the membrane is done by carrier proteins that are embedded in the membrane. ATP molecules (energy molecules) directly contribute to changing the shape of these carrier proteins, allowing them to open and close to let molecules in and out.
Any factors that can compromise the integrity of the carrier proteins will affect the movement of materials using this method. For example, high temperatures can cause the proteins to denature, stopping the movement of materials against the concentration gradient.
Lots of cells carry out some form of cytosis - the bulk movement of molecules into or out of the cell, through the cell membrane.
|
Endocytosis
The bulk movement of molecules into the cell.
White blood cells do endocytosis to engulf potentially harmful materials in our bodies in order to destroy them. The movement of molecules into the cell requires energy! There are two types of endocytosis: |
Exocytosis
The removal of bulk materials out of the cell. Secretory cells, such as cells found in our stomach, do exocytosis to release mucus which protects our stomach from the acid used to digest food.
The movement of molecules out of the cell requires energy! |
Enzymes
Enzymes are biological catalysts (natural speeder-uppers) that are able to facilitate reactions that break down molecules (catabolic reactions) or build them up (anabolic reactions).
-
Enzyme structure
-
How they work
-
Factors affecting enzymes
-
Examples
<
>
|
Enzymes are made up of proteins - different proteins come together to create different enzymes. The proteins fold up into complex, specific shapes that allow only one particular type of molecule to fit in them. This means they are specific. They work specifically on one type of molecule.
|
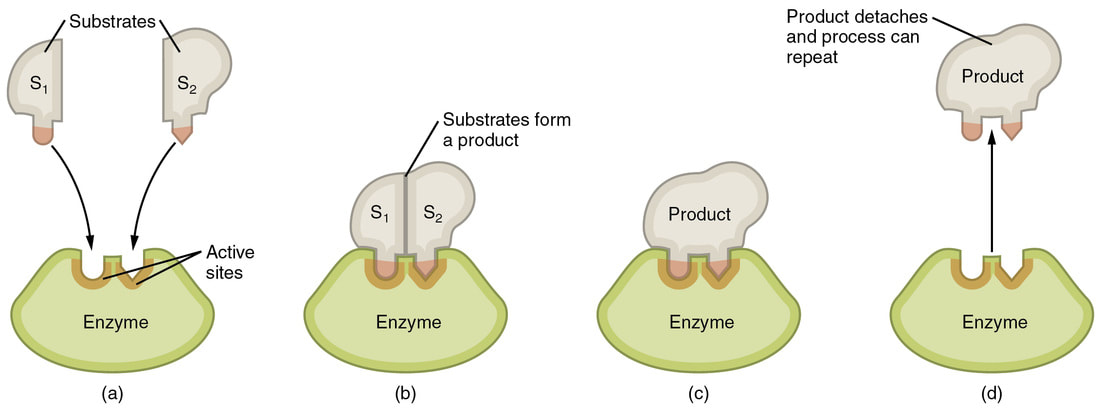
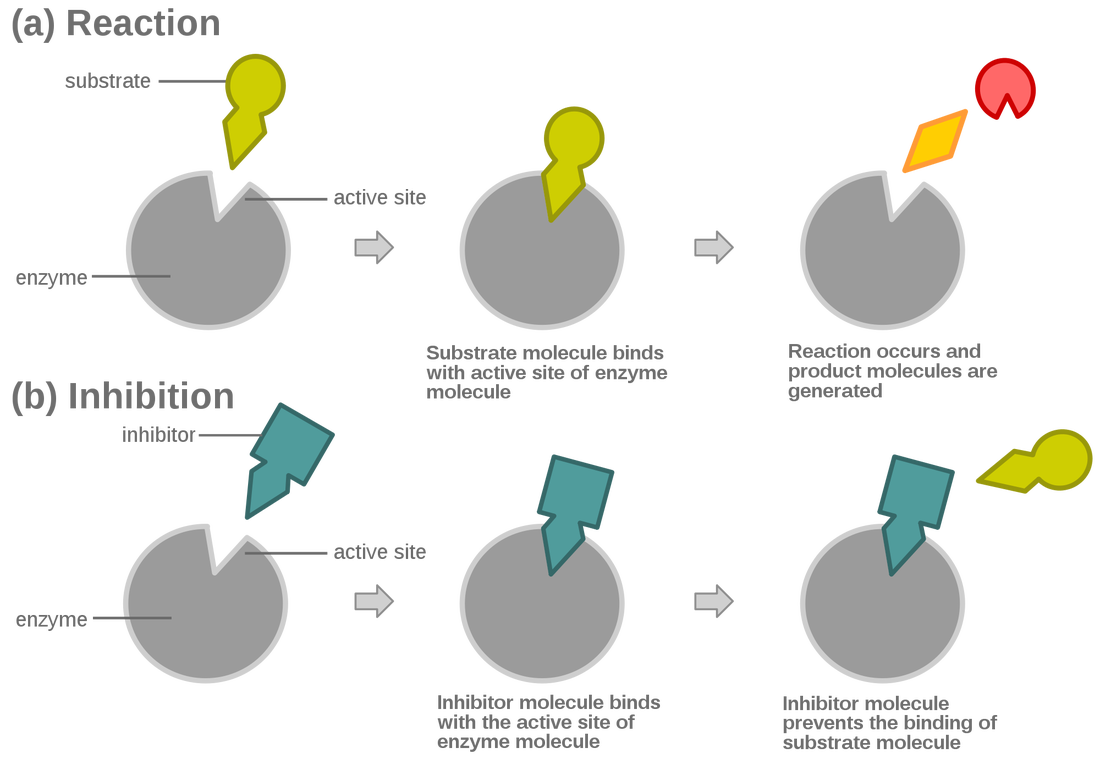
It is easier to picture enzymes as pacmans. The yellow part is the folded up protein and the gap is the active site. This area is where the specific type of molecule, called a substrate, fits so that it can be broken down or joined with another substrate.
|
the way enzymes work can be described using the induced fit model
|
Anabolic (building up) reactions
The substrates enter the active site (only those substrates can fit, not any other types) and undergo a reaction with help of the enzyme. They are then released as a completely new product. A common example of this type of reaction is DNA Polymerase (enzyme) joining okazaki fragments together (building up reaction)
|
Catabolic (breaking down) reactions
The substrate enters the active site and undergo a reaction with the help of the enzyme. There are then two or more products that are released by the enzyme. A common example of this type of reaction is during digestion - proteins, carbohydrates and lipids are broken down into amino acids, glucose and glycerol+fatty acids, respectively.
|
|
Enzymes work by lowering the activation energy required for reactions to occur. Activation energy is the minimum energy needed to make a reaction happen. Enzymes lower this amount by providing an alternative pathway for the reaction that requires less energy to occur. This means reactions can happen faster! |
|
Enzymes will be unable to work if they become denatured. This is when the bonds holding the enzyme in its specific shape are broken, and the enzyme unravels, losing the functionality of its active site and therefore its ability to catalyze any reactions. Certain environmental factors, such as temperature, can cause enzymes to denature.
|
|
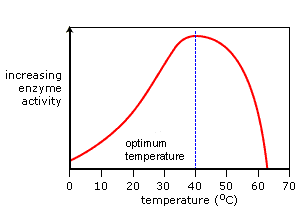
TEMPERATURE
As temperature increases, so does the rate of reaction (heat energy is a source of energy that enzymes use to catalyze reactions), until a certain temperature is reached (around 40 degrees Celsius) where the enzymes will start to denature. Once this happens, there will be a very small amount of (or no) enzymes and all reactions cease. Just before the point of denaturation is the 'optimum temperature' where the rate of reaction will be the highest. In cold temperatures, enzymes work slower due to the lack of energy they can get (lack of heat). |
|
PH
Enzymes have different 'optimum pH's - digestive enzymes work best in acidic solutions (such as pepsin, works best in a pH of 1-2). Other enzymes work best in neutral solutions etc. When the enzyme is in a pH that is classed as 'extreme' either side of its optimum pH, the enzyme will denature. |
|
INHIBITORS Inhibitors are molecules that basically get in the way. They bind to the active site, making it impossible for the correct substrate to get in there so a reaction can happen! Many poisons act as inhibitors for enzymes. When enzymes have an inhibitor in their active sites, the result would be a build up of the substrate! |
|
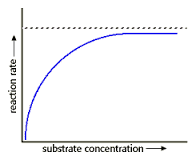
SUBSTRATE CONCENTRATION
The more substrate present, the faster the rate of reaction until a certain point is reached. This point is when all the enzymes are catalyzing reactions at their optimum rate - there is still more substrate to undergo a reaction but not enough enzymes to do so - therefore it's reached the fastest possible reaction rate. |